Introduction
Neurodegeneration research frequently examines protein misfolding, aggregation, and dysregulated proteostasis as shared molecular features across multiple disorders. Experimental literature commonly highlights amyloid precursor protein (APP), amyloid beta (Aβ), tau, α-synuclein, and TAR DNA-binding protein 43 (TDP-43) as proteins of interest because altered expression, processing, localization, or aggregation of these factors is associated with cellular stress responses, synaptic dysfunction, and inflammatory signaling in model systems.
Buntanetap is a small-molecule translational inhibitor investigated for its ability to modulate the synthesis of multiple proteins implicated in neurodegeneration research. Reported studies describe a mechanism in which buntanetap interacts with an iron-responsive element (IRE) within the untranslated region of specific mRNAs, influencing translation efficiency. IREs are regulatory RNA sequences that can bind iron-regulatory proteins (IRPs), thereby coupling iron homeostasis to gene expression control. In this context, buntanetap has been evaluated for effects on translation of targets such as APP and α-synuclein in experimental settings.
Mechanistic Focus: IRE-Linked Translational Regulation
A central hypothesis in the published research is that buntanetap’s binding to IRE-containing mRNA regions alters translation of select transcripts, which may reduce the production of proteins that participate in downstream aggregation pathways and stress signaling. This approach differs from strategies that target mature protein assemblies directly, as it is positioned upstream at the level of mRNA translation. Experimental work has assessed changes in protein abundance and related pathway markers following buntanetap exposure in cellular and animal model systems.
Figure 1. Buntanetap Chemical Structure
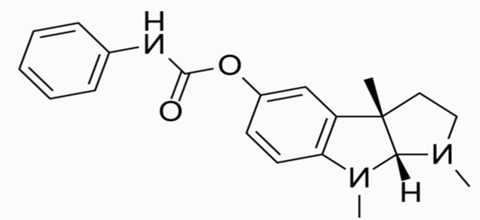
Research Findings Reported Across Model Systems
Across published preclinical and translational studies, buntanetap has been evaluated for its effects on multiple protein targets relevant to neurodegeneration research, including APP-related pathways and α-synuclein-associated aggregation models. Reported endpoints commonly include protein abundance measurements, inflammatory marker profiling, and readouts related to axonal or synaptic integrity in experimental systems. These studies generally frame buntanetap as a tool for probing how coordinated reductions in several neurotoxic proteins may reshape downstream cellular pathways.
TDP-43 as an Additional Translational Target Context
TDP-43 is a multifunctional RNA/DNA-binding protein involved in RNA processing, transport, and stress granule dynamics. A substantial body of literature describes pathological contexts in which TDP-43 mislocalization and aggregation are observed in neurodegeneration research. Genetic variation in TARDBP and related regulatory mechanisms have been studied for their association with altered TDP-43 handling, including changes in nucleocytoplasmic distribution and perturbations of axonal transport processes in experimental models.
Published investigations have also examined whether buntanetap influences TDP-43-related readouts. In these reports, the focus is typically on measuring TDP-43 abundance and associated pathway markers in research settings, while preserving the distinction between experimental observations and broader disease-level interpretations.
Figure 2. Experimental schematic of TDP-43–associated cellular pathways in motor neuron research models
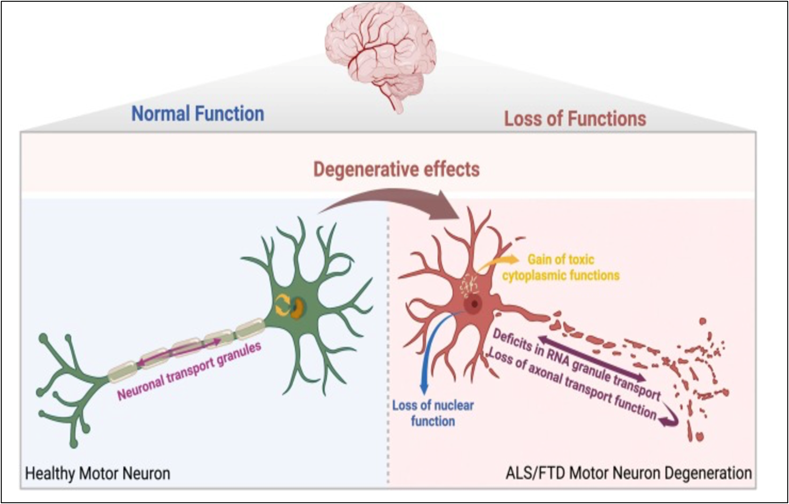
Research Scope and Ongoing Investigation
Overall, buntanetap is discussed in the literature as a research-stage small molecule used to investigate translational control mechanisms that may affect multiple proteins implicated in neurodegeneration-associated pathways. Current publications emphasize mechanistic questions such as target selectivity at the mRNA level, downstream pathway modulation, dose-response relationships in experimental systems, and how translation-linked interventions compare with alternative approaches aimed at aggregation, clearance, or inflammatory signaling.
References
- Babazadeh, A., Rayner, S. L., Lee, A., & Chung, R. S. (2023). TDP-43 as a molecular target in neurodegeneration research: focusing on motor neuron and frontotemporal degeneration models. Ageing Research Reviews, 102085.
- Fang, C., Hernandez, P., Liow, K., Chen, M., Zetterberg, H., Blennow, K., & Maccecchini, M. L. (2022). Translational inhibition of aggregation-associated proteins in experimental neurodegeneration models. Alzheimer’s & Dementia, 18, e068871.
- Fang, C., Hernandez, P., Liow, K., Damiano, E., Zetterberg, H., Blennow, K., et al. (2023). Translational regulation of multiple neurotoxicity-associated proteins in laboratory research systems. The Journal of Prevention of Alzheimer’s Disease, 10(1), 25–33.
- Forman, M. S., Trojanowski, J. Q., & Lee, V. M. (2007). TDP-43: a novel proteinopathy associated with neurodegenerative research models. Current Opinion in Neurobiology, 17(5), 548–555.
- Gaweda-Walerych, K., Sitek, E. J., Narożańska, E., & Buratti, E. (2021). Functional implications of Parkin downregulation in TDP-43 proteinopathies. Cells, 10(12), 3389.
- Johnson, B. (2022). Research perspectives beyond α-synuclein aggregation in neurodegeneration. Nature Biotechnology, 40(12), 1705–1707.
- Singh, A., Kukreti, R., Saso, L., & Kukreti, S. (2019). Oxidative stress as a regulatory factor in neurodegeneration-related molecular pathways. Molecules, 24(8), 1583.
The information presented in this article is provided solely for scientific, educational, and laboratory reference purposes. Any products or materials referenced are intended exclusively for in-vitro laboratory research use and are not intended for human or animal use, including diagnosis, treatment, mitigation, or prevention of any disease. No content herein should be construed as medical, clinical, or therapeutic guidance.


